Why All The Hate Towards The Alkaline Diet?
One of the best diets for longevity and staving off disease is the alkaline diet. We should be eating 80 percent alkaline and 20 percent acidic foods. Unfortunately most people have that ratio reversed, causing a pH imbalance and promoting the growth of illness and disease.
A 2012 review published in the Journal of Environmental Health found that the alkaline diet can protect healthy cells and balance essential mineral levels, thereby reducing morbidity and mortality from numerous chronic diseases and ailments.
This naturally leads me to ask why so many experts are now saying that the acid/alkaline diet principle is a mere theory, or unscientific bunk. The underpinning of their argument is that the pH of our blood is already tightly regulated by a complex system of buffers continuously working to maintain a range of 7.35 to 7.45, which is slightly alkaline.
One particular dietitian claims that there’s absolutely zero evidence that an alkaline diet ‘alkalizes’ anything. That’s an astounding dogmatic statement, despite a growing body of research identifying acidity as a very real phenomenon.
Here’s an entire page of scientific studies that tell a complete story. It’s important we get this subject right. Let’s examine why this diet really works!


Acid/Alkaline Principles
A pH value is is short for the potential of hydrogen (a measure of acidity or alkalinity of our body’s fluids and tissues). It’s measured on a scale from 0 to 14, where the higher the acidic a solution is, the lower its pH. The more alkaline, the higher the number pH value is. A value of around 7 is considered neutral, but the human body needs this value to be slightly alkaline at about 7.4.
Your bodily pH is indeed affected by the mineral density of the foods you eat, or in other words, the quality of your foods; an issue few experts will speak about.
There is no debate that the body has a tight buffering system, but it should also not be out of the realm of possibility that this system can be affected by an outside source such as food. Food gives us energy, phytochemicals, antioxidants, anti-inflammatories and, potentially, anti-cancer agents. So why should it not affect how our body regulates its pH balance?
The February 2015 issue of the journal Integrative Medicine provides one of the best overviews I’ve seen to tackle this subject. It identifies the difference between acidosis (the process leading to acidemia) and acidemia itself (defined as a blood pH of less than 7.35, or an actual change in blood pH). Acidemia would show up in advanced diseases like kidney and lung failure, but is an event unlikely to occur as the body has multiple mechanisms for ensuring a very stable blood pH.
Acidosis only becomes acidemia when bodily measures become overwhelmed with the constant pressure of too many acids. So while acidosis will not affect blood pH, the pH in the cells and intracellular space becomes more acidic, causing disruption of enzyme function, loss of insulin sensitivity, and cellular metabolic adaptations.
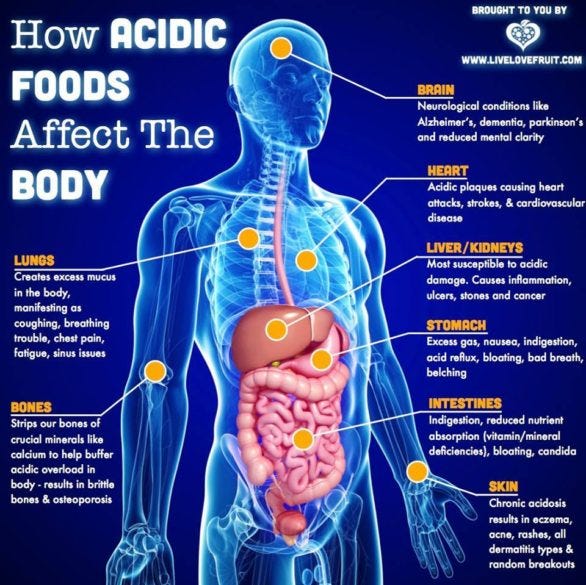
Healthy kidneys and lungs will do their best to get rid of almost all of excessive acidity. But keep pushing an unhealthy diet for too long and these systems start to fail. The mineral content from 3 g of bone is needed to neutralize 1 g of acid, and this turns out to be a seriously underrecognized cause of osteoporosis. (Integr Med)
The primary sources of acidity in the diet are sulfur-containing amino acids, salt, and phosphoric acid in soft drinks (IMCJ 13.6). Salt is everywhere, that’s no great mystery. But research also reveals that we consume far more phosphorus than the recommended dietary allowance (RDA) (700 mg/d for adults). Some individuals’ daily phosphorus intake exceeds even an adult’s tolerable upper limit (4000 mg/d). To avoid this, you must also avoid processed foods, virtually all of which are laden with phosphate/salt-containing food additives. (Also Nutrients 2017, 9(6), 538)
Contrarily, alkalinity supplies important nutrients that help stop rapid degeneration of tissues, bone mass and disease.
Fruits and vegetables protect against decreased bone strength and muscle wasting; a condition known as sarcopenia by balancing the ratio of minerals that are important for bones and maintaining lean muscle mass, including calcium, magnesium and phosphate.
Another benefit is a reduction in inflammation as well as a beneficial increase in growth hormone production improving cardiovascular health and helping against high cholesterol, hypertension (high blood pressure), kidney stones, stroke and even memory loss.
Other nutrients identified as necessary for proper calcium utilization and bone quality are vitamins K, K2, C and B complex, magnesium and silica. Vitamins C and B complex, zinc, selenium, boron and copper and strontium have also shown research promise for bone strength (Endocrinol Nutr).
Also realize that some foods are “acidic” or “alkaline” by nature (composition), versus their potential effect on the body. Lemon juice and tomatoes, for example, are acidic. But when ingested, they promote alkalinity. Even when eating healthy acidic foods — such as poultry, whole grains and eggs — this is not necessarily detrimental but may need to be combined with alkaline-forming foods (more fruits and vegetables) to make the net effect of the meal either neutral or alkaline.
Different systems of the body also maintain different pH levels, with some more tightly regulated than others. For example, the stomach maintains a very acidic pH at 1.2 to 3.0 so that it can break down dietary protein and kill ingested pathogens. Fluid inside cells can range between slightly acidic and slightly alkaline, but an alkaline environment within cells (achievable through an alkaline-promoting diet), improves cellular function. (foodandnutrition.org)
What Are Acid/Alkaline Forming Foods?
In simple terms, your body goes through a complicated process to make sure your blood pH stays balanced and won’t rob nutrients from elsewhere IF you eat a balanced diet to support your body’s natural processes!
‘The body functions at its best when the pH of its internal bio-chemical environment, measured as a whole, is equal to 7.39, meaning slightly alkaline. Christopher Vasey, Expert on the Alkaline Diet
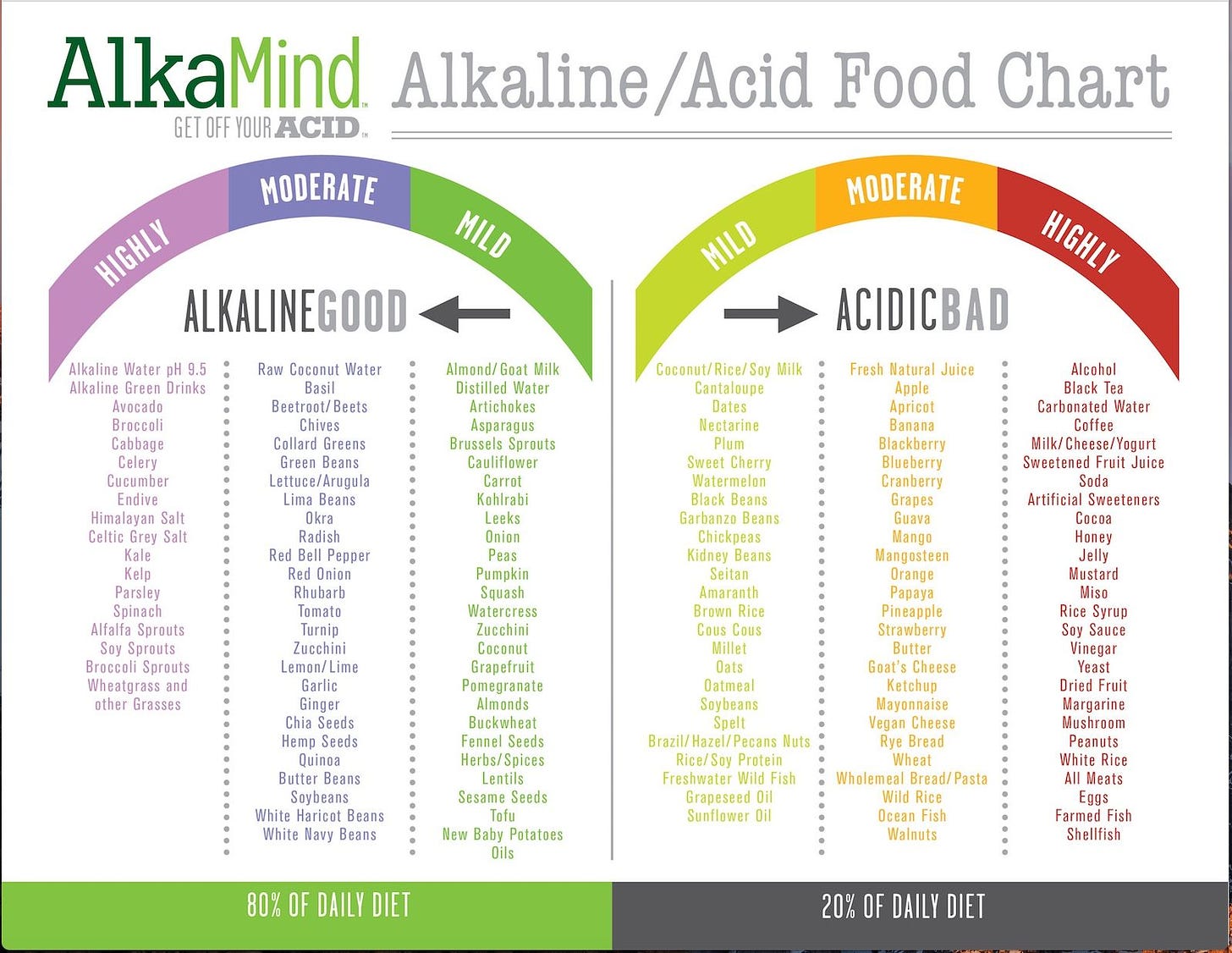
Understanding how to achieve alkalinity is not overly complicated. Simply have a look at your plate: is 70-80 percent of it made up of alkaline foods? And does the remaining 20-30 percent of acid forming foods belong to the category of natural, slightly or highly acidifying foods?
Generally speaking, most vegetables and fruits have an alkaline-forming effect on your body fluids, while most grains, animal foods, and highly processed foods have an acid-forming effect.
The following list indicates the difference between the two when digested and assimilated into your system. Any oils on the list should be expeller pressed, not commercially produced.
Although the chart rates foods good and bad, mild to moderate acidic foods are not inherently bad, but only out of moderation. So stick with more alkaline foods, as the chart suggests:
“The available research makes a compelling case that diet-induced acidosis, not diet-induced acidemia, is a real phenomenon, and has a significant, clinical, long-term pathophysiological effect that should be recognized and potentially counterbalanced by dietary means.” Published In: British Journal of Nutrition; Br J Nutr. 2010 Apr;103(8):1185-94.
So eat an alkalizing diet rich in fruits and vegetables in order to avoid diet-induced metabolic acidosis and preserve bone mass. (Eur J Nutr)